Pharma’s digital challenge: Medical mis-and disinformation and unreported adverse events

The rapid growth of social media and digital communication platforms have fundamentally transformed how biopharmaceutical companies communicate and interact with patients. According to the latest figures on social media compiled by Datareportal, with over 4.8 billion users globally, social networking platforms can provide brands instantaneous access to feedback from a diverse audience of consumers.
In this blogpost, Resolver will cover two of the most significant digital threats facing pharmaceutical companies as they increase their presence online. The blog also outlines how major brands in this sector employ cutting-edge technology and network intelligence drawn from teams of experienced human analysts to detect risk signals embedded in online discourse and ensure they are always the first to know, and the first to act.
Medical mis-and disinformation
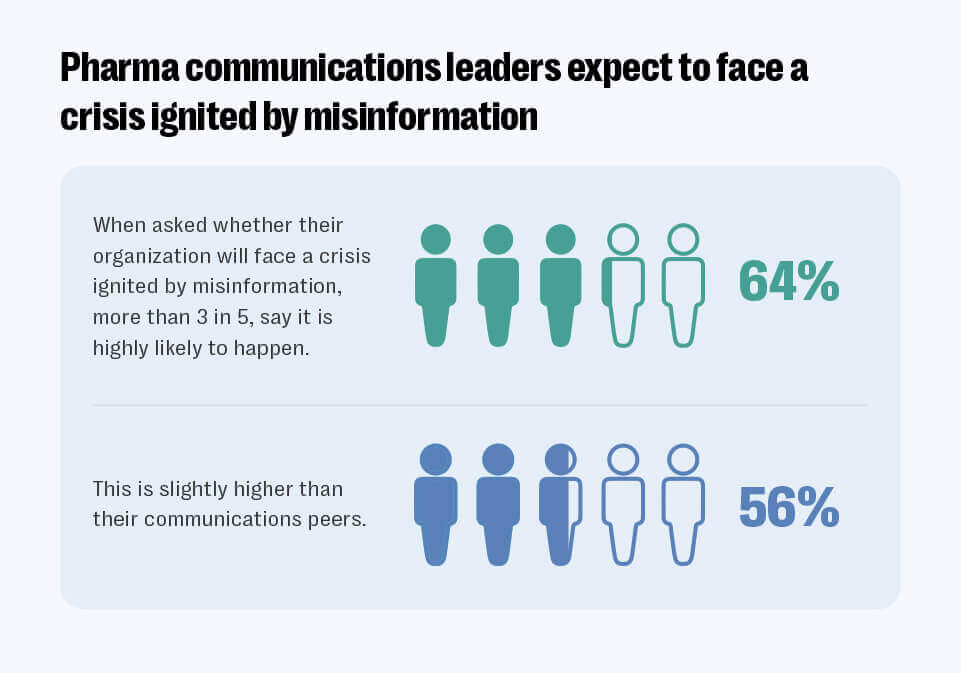
The widespread amplification of medical mis-and disinformation via social media platforms represents a significant threat to major pharma brands in 2023 according to a Resolver in-house survey of 100 communication and pharmaceutical industry professionals in the US, Canada and Europe. The results of the survey conducted as part of the Communication Leaders Risk Survey: Pharma Edition revealed that more than three in five (64%) of respondents believed it was highly likely their business will face a crisis ignited by misinformation.
The “infodemic” of false and misleading narratives proliferated online during the recent COVID-19 pandemic revealed how medical mis-and disinformation can severely undermine public health initiatives by causing decreased uptake of health care resources, delays in patients seeking care, vaccine hesitancy, medication non-compliance contributing towards increased hospitalization and patient mortality.
The increased public anxiety around a public health crisis can also be leveraged by malicious actors to amplify content aimed at creating doubt and uncertainty over the efficacy of products associated with a particular brand. Alarmingly, this risk can also extend to targeted campaigns directed at specific C-suite executives working at such firms. A notable example of such targeted harassment is provided by a series of manipulated images and videos using the likeness of a Chief Executive Officer at a prominent pharma brand to promote conspiratorial and anti-Vaxx narratives online.
Reiterating the gravity of this threat, Emma Durant, Strategic Customer Success Manager at Resolver, a Kroll business warns ‘as we saw with two of our major clients in this sector at the start of the pandemic, mis-and disinformation, if widespread, can quickly overtake the truth online, meaning that potential consumers will have a false impression of how the drug works’. Adding that, ‘this can lead to consumers deciding against the use of the drug or even boycotting the company if they feel that they have been involved in activity which they do not want to align themselves with.’
The speed with which unsubstantiated rumors can spread on social networks necessitates that brands in this sector adopt a quick and reflexive approach towards communicating with consumers via their social channels.
‘‘Finding out the narrative as quickly as possible is key, so that the company can prepare a strategic response’ advises Emma Durant, in this regard access to up-to-date network intelligence is integral as it allows brands to ‘understand where the misleading information started, so that they can quickly work with the relevant social platform to try to close it down’, adding that ‘putting out the fire as close to the spark is vital in making sure it doesn’t spread’.
“Finding out the narrative as quickly as possible is key, so that the company can prepare a strategic response. Putting out the fire as close to the spark is vital in making sure it doesn’t spread.“
Emma Durant
Strategic Customer Services Manager,
Resolver, a Kroll business
Unreported adverse events
After medical mis-and disinformation, unreported adverse events on social media represent another significant source of operational and reputational harm for major pharma brands, with non-compliance likely to result in audits, fines, regulatory sanctions, legal challenges, and public relations crises.
‘The operational risk is major’ warns Emma Durant, Strategic Customer Success Manager at Resolver, ‘brands can be fined by the Food and Drug Administration (FDA) or European Medicines Agency (EMA) if they miss the reporting of an adverse event on their online channels, within the specified time period.’ Unwanted media coverage can cause further damage ‘if it was reported in the media that they had been fined, it would no doubt have some impact on their overall reputation and potentially share price depending on the size of the fine’ she added.
“The operational risk is major. brands can be fined by the Food and Drug Administration (FDA) or European Medicines Agency (EMA) if they miss the reporting of an adverse event on their online channels, within the specified time period.“
Emma Durant
Strategic Customer Services Manager,
Resolver, a Kroll business
In July 2023, Reuters reported on the EMA’s decision to launch an investigation into two products produced by a Danish pharma brand related to diabetes and weight loss. The investigation was launched on the basis of just three cases of adverse effects reported by patients to a national health regulator, and led to shares of the drugmaker falling by 1% due to the subsequent media coverage.
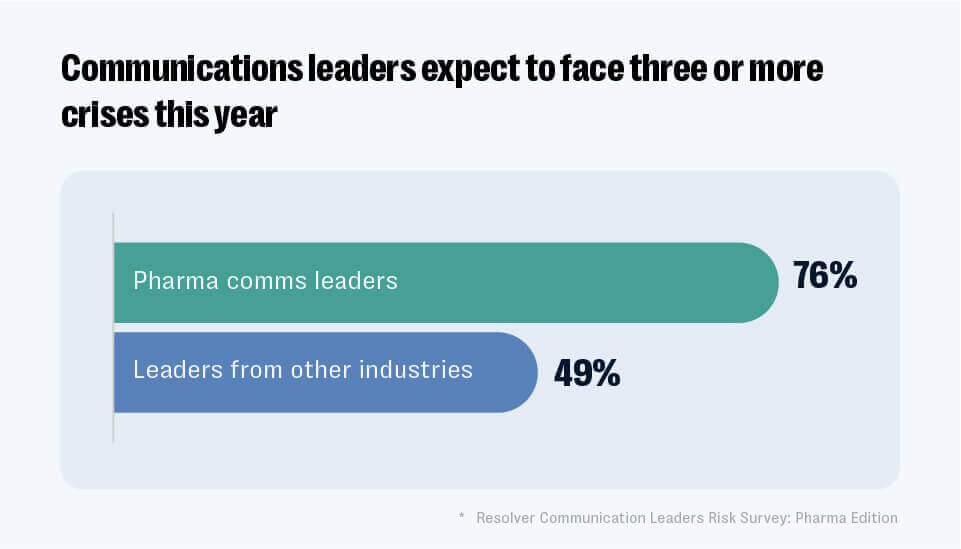
Despite the difficulties of processing and verifying such large quantities of safety data, the risks of falling afoul of compliance regulations can be severe. The virality factor inherent in the online informational environment has created an incessant cycle of accelerated risks forcing major pharma brands into a state of constant vigilance. According to insights drawn from the Communication Leaders Risk Survey: Pharma Edition more than three-quarters (76%) of pharma communicators expect to face three or more crises per year versus less than half (49%) of their peers in other industries.
In theory, social networking platforms can significantly enhance a brands pharmacovigilance practice by increasing connections between companies and healthcare consumers that can facilitate and enable safety reporting through direct patient and consumer interaction. In practice, however, verifying this safety data can prove challenging, particularly given the issue of confirming the “identifiability” of both reporter and patient. This refers to the responsibility of a brand to verify the credibility and contact details of the reporter and determine whether the feedback originates from a real person.
Adverse event data amplified via social media often contains personal identifiable information related to the patient and the reporter, as a result brands processing this data will need to ensure their collection, analysis and storage of this data is in compliance with applicable data protection laws and supported by transparent processes designed to ensure personal data protection.
The responsibility for building out such capacities will likely fall on the shoulders of in-house social media managers at major pharma brands, diverting their energy and resources away from their day-to-day tasks.
Conclusion
Leveraging the wealth of open-source data available across platforms allows brands to implement patient-centric models built upon monitoring, analyzing and reporting compliance data from consumers in real-time. However, the adoption of that approach could exacerbate their operational risks, particularly within the context of medical mis-and disinformation targeting their products and challenges posed by adverse event reporting.
By partnering with Resolver, a Kroll business’s Compliance and Adverse Event detection solution, pharma brands can minimize the disruption caused by such digital threats. With 100% guaranteed adverse event detection our algorithms and teams of analysts work around the clock to detect risk signals and adverse event data embedded in online discussions and verify the motives, networks and actors behind them.



